Summer is here and once more fires are burning woods and forests in the northern hemisphere from Greece to USA and unfortunately most of the times because someone irresponsible and crazy enough “thought” that he/she will benefit from it.
In this article I am going to discuss the chemical reactions that occur while wood is burnt and the gasses that release into the atmosphere.
Theory
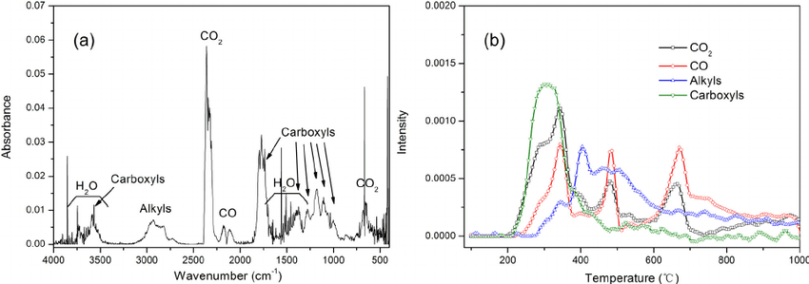
Once the wood gets to about 150°C/302°F, the cellulose in it starts to decompose (a process called Pyrolysis) and releases VOCs gases. The burning of the VOCs provides most of the flames you see. These volatile gases are compounds made up of the elements Hydrogen (H), Carbon (C) and Oxygen (O). Once the gases are released the rest of the fuel material forms char, which is nearly pure Carbon and ash, which is all the unburnable minerals in the fuel (like calcium, potassium, etc).
The chemical composition of the wood is 6C10H15O7 and when heat is applied then it is converted into C50H10O which is Char + the 10CH2O gas, did you recognize this gas, it is Formaldehyde.
6C10H15O7 + HEAT = C50H10O + 10CH2O
Once the volatile gases reach about 260°C/500°F the compound breaks apart and a second chemical reaction called Gasification occurs with the oxygen in the atmosphere to create water, carbon dioxide and other waste products. This reaction gives off a lot of heat energy (exothermic), which then feeds further chemical reactions, so fire is a self-perpetuating reaction, as long as there is fuel and oxygen present.
6CH2O + 3O2 = 6H2O + 2CO2 + 2CO + 2C + HEAT
Reality
Of course, a forest or a wood pallet for domestic burners it is not made of 100% pure 6C10H15O7 wood cellulose because every tree has different chemical compounds in it, as a result there are many other countless chemicals that when they get burned they release a ton of other chemicals into the atmosphere.
For example, the wood pallets most of the times are treated with Methyl Bromide for heat-resistant or even Chromated Copper Arsenate. What toxic substances will be liberated to the environment when the As2CrCuO9 or CH3Br get burnt with the wood?
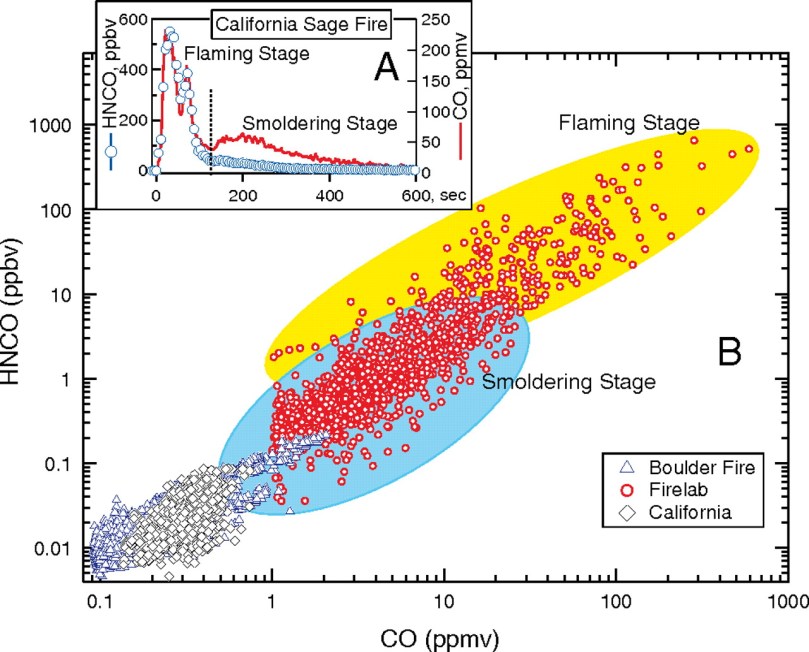
The same happens with natural wood like pine wood which is rich in pine oil and compounds like Terpineol C10H18O, Limonene C10H16, etc. For example, a NOAA study found Isocyanic Acid HNCO in the smoke from the Fourmile Canyon wildfire west of Boulder, Colorado in 2010. When different species of trees are burnt different chemicals occur. Temperature and soil also plays an important role.
Discover more from See The Air
Subscribe to get the latest posts sent to your email.


[…] Wood Fire & Chemical Reactions Why does charcoal burn hotter then the wood it is made from? byu/WarnikOdinson inaskscience […]
LikeLiked by 1 person
[…] sneaky little particles are causing headaches for anyone trying to keep the air squeaky clean. See The Air points out that while VOCs add a cool visual flair to your fire, they’re not doing any favors […]
LikeLiked by 1 person